READ: Chemical Formulas
1. Chemical Symbols
Chemical symbols are one or two letters used to represent every element. Some common examples of chemical symbols would be H for hydrogen, O for oxygen, C for carbon, N for nitrogen, and Na for sodium. The first letter of the symbol is always capitalized. If the symbol contains two letters the second letter is lowercase.
The symbols for every element are shown on the periodic table. Click HERE to open a basic periodic table. You can see the name of each element along with its symbol.
Compounds are combinations of two or more elements.
A chemical formula is an expression that shows the elements in a compound and the relative proportions of those elements. In a chemical formula, the elements in a compound are represented by their chemical symbols, and the ratio of different elements is represented by subscripts. A subscript is a number that is smaller than the normal type and is set slightly below.
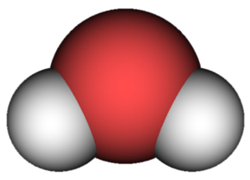
Water is a compound. Each water molecule contains two hydrogen atoms and one oxygen atom. Therefore, the chemical formula for water is:
H2O
The subscript 2 after the H shows that there are two atoms of hydrogen in the molecule. The O for oxygen has no subscript. When there is just one atom of an element in a molecule, no subscript is used in the chemical formula. A picture of a water molecule is shown below. The red circle represents the single oxygen atom. The grey circles represent the two hydrogen atoms.