READ: Chemical Formulas
| Site: | Mountain Heights Academy OER |
| Course: | Integrated Science 8 Q1 |
| Book: | READ: Chemical Formulas |
| Printed by: | Guest user |
| Date: | Friday, 25 July 2025, 4:38 PM |
1. Chemical Symbols
Chemical symbols are one or two letters used to represent every element. Some common examples of chemical symbols would be H for hydrogen, O for oxygen, C for carbon, N for nitrogen, and Na for sodium. The first letter of the symbol is always capitalized. If the symbol contains two letters the second letter is lowercase.
The symbols for every element are shown on the periodic table. Click HERE to open a basic periodic table. You can see the name of each element along with its symbol.
Compounds are combinations of two or more elements.
A chemical formula is an expression that shows the elements in a compound and the relative proportions of those elements. In a chemical formula, the elements in a compound are represented by their chemical symbols, and the ratio of different elements is represented by subscripts. A subscript is a number that is smaller than the normal type and is set slightly below.
Water is a compound. Each water molecule contains two hydrogen atoms and one oxygen atom. Therefore, the chemical formula for water is:
H2O
The subscript 2 after the H shows that there are two atoms of hydrogen in the molecule. The O for oxygen has no subscript. When there is just one atom of an element in a molecule, no subscript is used in the chemical formula. A picture of a water molecule is shown below. The red circle represents the single oxygen atom. The grey circles represent the two hydrogen atoms.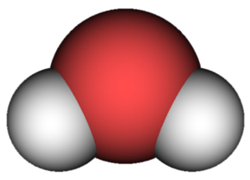
Sources:
Water Molecule, Sulfuric Acid Molecule, ck12 Chemical Formulas, ck12 Chemical Symbols
2. Chemical Equations
Look at this rusty bike wheel. It has been left outside in damp weather too many times, so the iron in the metal parts has rusted. Iron rusts when it combines with oxygen in the air. Iron rusting is an example of a chemical reaction. In a chemical reaction, substances change into entirely different substances. For example, the iron in the bike and the oxygen in the air have changed into rust. How could you represent this reaction, besides just describing it in words? Scientists use a standard method to represent a chemical reaction, called a chemical equation.
What Is a Chemical Equation?
A chemical equation is a shorthand way to sum up what occurs in a chemical reaction. The general form of a chemical equation is:
- Reactants → Products
The reactants in a chemical equation are the substances that begin the reaction, and the products are the substances that are produced in the reaction. The reactants are always written on the left side of the equation and the products on the right. The arrow pointing from left to right shows that the reactants change into the products during the reaction.
The general equation for the reaction in which iron rusts is shown below as an example:
- Iron + Oxygen → Iron Oxide
Using Chemical Symbols and Formulas
When scientists write chemical equations, they use chemical symbols and chemical formulas instead of names to represent reactants and products. Look at the chemical reaction illustrated below. In this reaction, carbon reacts with oxygen to produce carbon dioxide. Carbon is represented by the chemical symbol C. The chemical symbol for oxygen is O, but pure oxygen exists as diatomic (“two-atom”) molecules, represented by the chemical formula O2. A molecule of the compound carbon dioxide consists of one atom of carbon and two atoms of oxygen, so carbon dioxide is represented by the chemical formula CO2.
The chemical equation for this reaction is:
- C + O2 → CO2
3. Conservation of Mass
If you build a campfire like this one, you start with a big pile of logs. As the fire burns, the pile of logs slowly shrinks. By the end of the evening, all that’s left is a small pile of ashes. What happened to the matter that you started with? Was it destroyed by the fire?
Where’s the Matter?
It may seem as though burning destroys matter, but the same amount, or mass, of matter still exists after a campfire as before. Look at the sketch in Figure below. It shows that when wood burns, it combines with oxygen and changes not only to ashes but also to carbon dioxide and water vapor. The gases float off into the air, leaving behind just the ashes. Suppose you had measured the mass of the wood before it burned and the mass of the ashes after it burned. Also suppose you had been able to measure the oxygen used by the fire and the gases produced by the fire. What would you find? The total mass of matter after the fire would be the same as the total mass of matter before the fire.
Law of Conservation of Mass
This burning campfire example illustrates a very important law in science: the law of conservation of mass. This law states that matter cannot be created or destroyed. Even when matter goes through a physical or chemical change, the total mass of matter always remains the same.
4. Balancing Equations
Is It Balanced?
All chemical equations, like equations in math, must balance. This means that there must be the same number of each type of atom on both sides of the arrow. That’s because matter is always conserved in a chemical reaction. This is the law of conservation of mass.
Look at the equation above for the reaction between carbon and oxygen in the formation of carbon dioxide. Count the number of atoms of each type. Are the numbers the same on both sides of the arrow? The answer is yes, so the equation is balanced.
Coefficients
Let’s return to the chemical reaction in which iron (Fe) combines with oxygen (O2) to form rust, or iron oxide (Fe2O3). The equation for this reaction is:
4Fe+ 3O2 → 2Fe2O3
This equation illustrates the use of coefficients to balance chemical equations. A coefficient is a number placed in front of a chemical symbol or formula that shows how many atoms or molecules of the substance are involved in the reaction. From the equation for rusting, you can see that four atoms of iron combine with three molecules of oxygen to form two molecules of iron oxide.
Q: Is the equation for the rusting reaction balanced? How can you tell?
A: Yes, the equation is balanced. You can tell because there is the same number of each type of atom on both sides of the arrow. First count the iron atoms. There are four iron atoms in the reactants. There are also four iron atoms in the products (two in each of the two iron oxide molecules). Now count the oxygen atoms. There are six on each side of the arrow, confirming that the equation is balanced in terms of oxygen as well as iron.
5. Summary and Vocabulary
Summary
- Scientists use chemical equations to summarize what happens in chemical reactions. Reactants are placed on the left side of the equation and products are placed on the right. An arrow is used to indicate the direction in which the reaction occurs. Plus signs (+) are placed between multiple reactants or products.
- In chemical equations, reactants and products are represented by chemical symbols and formulas. Numbers called coefficients are placed in front of the symbols and formulas to show how much of each substance is involved in the reaction.
- Chemical equations must be balanced. A balanced equation has the same number of each type of atom on both sides of the equation.
- Burning and other changes in matter do not destroy matter. The mass of matter is always the same before and after the changes occur.
- The law of conservation of mass states that matter cannot be created or destroyed.
Vocabulary
- Chemical symbols: one or two letters used to represent every element.
- Compounds: combinations of two or more elements.
- Chemical formula: an expression that shows the elements in a compound and the relative proportions of those elements.
- subscript: a number that is smaller than the normal type and is set slightly below.
- chemical equation: Symbolic representation of a chemical reaction.
- law of conservation of mass: Law stating that matter cannot be created or destroyed in chemical reactions.