EXPLORE: Life on Earth
| Site: | Mountain Heights Academy OER |
| Course: | Earth Science Q1 |
| Book: | EXPLORE: Life on Earth |
| Printed by: | Guest user |
| Date: | Saturday, 26 July 2025, 3:01 PM |
Description
This book explains why Earth is uniquely habitable in our solar system
1. Why Earth Supports Life
The following screencast explains the key ingredients necessary for supporting life. Earth is unique in our solar system because it is the only planet known to be habitable. This lecture explains why, and explores several other places in our solar system life may, someday, be found to exist.
2. Goldilocks and the Greenhouse Effect
The Goldilocks Principle can be summed up neatly as "Venus is too hot, Mars is too cold, and Earth is just right." The fact that Earth has an average surface temperature comfortably between the boiling point and freezing point of water, and thus is suitable for our sort of life, cannot be explained by simply suggesting that our planet orbits at just the right distance from the sun to absorb just the right amount of solar radiation. Our moderate temperatures are also the result of having just the right kind of atmosphere. A Venus-type atmosphere would produce extremely hot conditions on our planet; a Mars atmosphere would leave us shivering in a Martian-type deep freeze.
Instead, parts of our atmosphere act as an insulating blanket of just the right thickness, trapping sufficient solar energy to keep the global average temperature in a pleasant range. The Martian blanket is too thin, and the Venusian blanket is way too thick! The 'blanket' here is a collection of atmospheric gases called 'greenhouse gases' based on the idea that the gases also 'trap' heat like the glass walls of a greenhouse do.
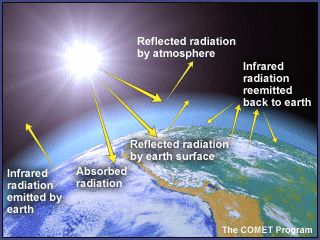
Sources
http://www.ucar.edu/learn/1_3_1.htm (Copyrighted, educational purposes permitted.)
3. Solar Radiation, Earth's Atmosphere, and the Greenhouse Effect
The sun radiates vast quantities of energy into space, across a wide spectrum of wavelengths.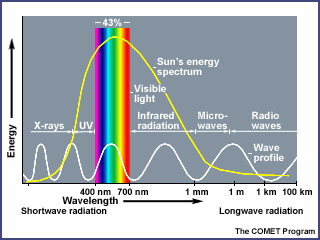
Source
http://www.ucar.edu/learn/1_3_1.htm (Copyrighted, educational use permitted)
4. Greenhouse Effect
Carbon dioxide CO2 is one of the greenhouse gases. It consists of one carbon atom with an oxygen atom bonded to each side. When its atoms are bonded tightly together, the carbon dioxide molecule can absorb infrared radiation and the molecule starts to vibrate. Eventually, the vibrating molecule will emit the radiation again, and it will likely be absorbed by yet another greenhouse gas molecule. This absorption-emission-absorption cycle serves to keep the heat near the surface, effectively insulating the surface from the cold of space.

Greenhouse Effect
Atmospheric scientists first used the term 'greenhouse effect' in the early 1800s. At that time, it was used to describe the naturally occurring functions of trace gases in the atmosphere and did not have any negative connotations. It was not until the mid-1950s that the term greenhouse effect started being used when discussing climate change. You will learn more about climate change later.
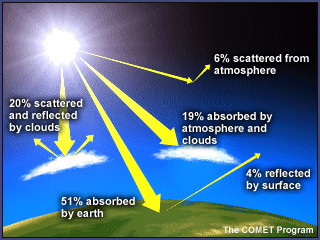
While the earth's temperature is dependent upon the greenhouse-like action of the atmosphere, the amount of heating and cooling are strongly influenced by several factors just as greenhouses are affected by various factors.
In the atmospheric greenhouse effect, the type of surface that sunlight first encounters is the most important factor. Forests, grasslands, ocean surfaces, ice caps, deserts, and cities all absorb, reflect, and radiate radiation differently. Sunlight falling on a white glacier surface strongly reflects back into space, resulting in minimal heating of the surface and lower atmosphere. Sunlight falling on a dark desert soil is strongly absorbed, on the other hand, and contributes to significant heating of the surface and lower atmosphere. Cloud cover also affects greenhouse warming by both reducing the amount of solar radiation reaching the earth's surface and by reducing the amount of radiation energy emitted into space.