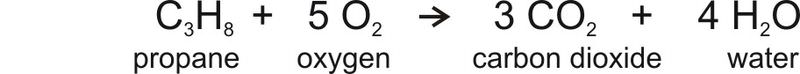READ: The Carbon Cycle
READ: The Carbon Cycle
Burning of fossil fuels, such as oil, releases carbon into the atmosphere. This carbon must be cycled - removed from the atmosphere - back into living organisms, or it stays in the atmosphere. Increased carbon in the atmosphere contributes to the greenhouse effect on Earth.
The Carbon Cycle
Carbon is a very important element to living things. As the second most common element in the human body, we know that human life without carbon would not be possible. Carbon occurs in many forms on Earth. The element moves through organisms and then returns to the environment. When all this happens in balance, the ecosystem remains in balance too.
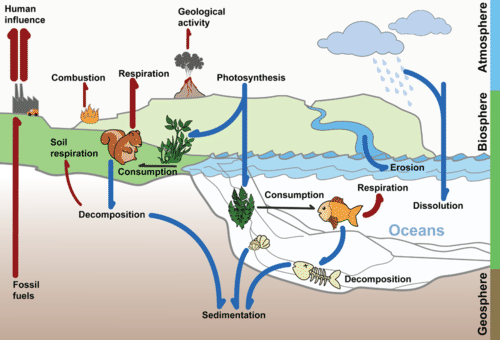
Flowing water can slowly dissolve carbon in sedimentary rock. Most of this carbon ends up in the ocean. The deep ocean can store carbon for thousands of years or more. Sedimentary rock and the ocean are major reservoirs of stored carbon. Carbon is also stored for varying lengths of time in the atmosphere, in living organisms, and as fossil fuel deposits. These are all parts of the carbon cycle, which is shown in the figure below.
The Carbon Cycle. Carbon moves from one reservoir to another in the carbon cycle. What role do organisms play in this cycle?
Why is recycling carbon important? Recall that carbon is the cornerstone of organic compounds, the compounds necessary for life. But do organisms make their own carbon? No. Carbon must be recycled from other living organisms, from carbon in the atmosphere, and from carbon in other parts of the biosphere.
Carbon in Carbon Dioxide
Carbon cycles quickly between organisms and the atmosphere. In the atmosphere, carbon exists primarily as carbon dioxide (CO2). Carbon dioxide cycles through the atmosphere by several different processes, including those listed below.
- Living organisms release carbon dioxide as a byproduct of cellular respiration.
- Photosynthesis removes carbon dioxide from the atmosphere and uses it to make organic compounds.
- Carbon dioxide is given off when dead organisms and other organic materials decompose.
- Burning organic material, such as fossil fuels, releases carbon dioxide.
- Carbon cycles far more slowly through geological processes such as sedimentation. Carbon may be stored in sedimentary rock for millions of years.
- When volcanoes erupt, they give off carbon dioxide that is stored in the mantle.
- Carbon dioxide is released when limestone is heated during the production of cement.
- Ocean water releases dissolved carbon dioxide into the atmosphere when water temperature rises.
- Carbon dioxide is also removed when ocean water cools and dissolves more carbon dioxide from the air.
Short Term Cycling of Carbon
The short term cycling of carbon begins with carbon dioxide (CO2) in the atmosphere.
Photosynthesis and Respiration
When plants do photosynthesis, the in carbon dioxide in the air combines with water and energy from sunlight and is transformed into organic carbon (food). The chemical equation for photosynthesis is:
Plants and animals also engage in the reverse of photosynthesis, which is respiration. In respiration, organic carbon in sugar is converted into energy. This releases carbon dioxide from the plants and animals. The chemical reaction for respiration is:
C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + useable energy
Photosynthesis and respiration are a gas exchange process. In photosynthesis, CO2 is converted to O2; in respiration, O2 is converted to CO2.
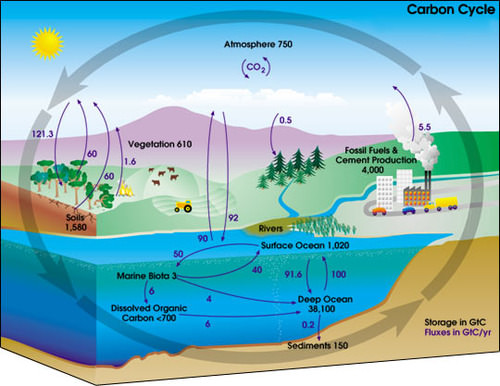
The carbon cycle shows where a carbon atom might be found. The black numbers indicate how much carbon is stored in various reservoirs, in billions of tons ("GtC" stands for gigatons of carbon). The purple numbers indicate how much carbon moves between reservoirs each year. The sediments, as defined in this diagram, do not include the ~70 million GtC of carbonate rock and kerogen.
Long-Term Carbon Cycling
Carbon Sinks and Carbon Sources
Places in the ecosystem that store carbon are reservoirs. Places that supply and remove carbon are carbon sources and carbon sinks, respectively. If more carbon is provided than stored, the place is a carbon source. If more carbon dioxide is absorbed than is emitted, the reservoir is a carbon sink. What are some examples of carbon sources and sinks?
- Carbon sinks are reservoirs where carbon is stored. Healthy living forests and the oceans act as carbon sinks.
- Carbon sources are reservoirs from which carbon can enter the environment. The mantle is a source of carbon from volcanic gases.
A reservoir can change from a sink to a source and vice versa. A forest is a sink, but when the forest burns it becomes a source.
The amount of time that carbon stays, on average, in a reservoir is the residence time of carbon in that reservoir.
Atmospheric Carbon Dioxide
Remember that the amount of CO2 in the atmosphere is very low. This means that a small increase or decrease in the atmospheric CO2 can have a large effect.
By measuring the composition of air bubbles trapped in glacial ice, scientists can learn the amount of atmospheric CO2 at times in the past. Of particular interest is the time just before the Industrial Revolution, when society began to use fossil fuels. That value is thought to be the natural content of CO2 for this time period; that number was 280 parts per million (ppm).
By 1958, when scientists began to directly measure CO2 content from the atmosphere at Mauna Loa volcano in the Pacific Ocean, the amount was 316 ppm (Figure below). In 2014, the atmospheric CO2 content crossed over 400 ppm.
The amount of CO2 in the atmosphere has been measured at Mauna Loa Observatory since 1958. The blue line shows yearly averaged CO2. The red line shows seasonal variations in CO2.
This is an increase in atmospheric CO2 of 40% since the before the Industrial Revolution. About 65% of that increase has occurred since the first CO2 measurements were made on Mauna Loa Volcano, Hawaii, in 1958.
Carbon in the Atmosphere
Though carbon can be found in ocean water, rocks and sediment and other parts of the biosphere, the atmosphere may be the most recognizable reservoir of carbon. Carbon occurs in various forms in different parts of the carbon cycle. Some of the different forms in which carbon appears are described in the table below. KEY: C = Carbon, O = Oxygen, H = Hydrogen
| Form of Carbon | Chemical Formula | State | Main Reservoir |
|---|---|---|---|
| Carbon Dioxide | CO2 | Gas | Atmosphere |
| Carbonic Acid | H2CO3 | Liquid | Ocean |
| Bicarbonate Ion | HCO3− | Liquid(dissolved ion) | Ocean |
| Organic Compounds | Examples: C6H12O6 (Glucose), CH4 (Methane) | Solid Gas | Biosphere Organic Sediments (Fossil Fuels) |
| Other Carbon Compounds | Examples: CaCO3 (Calcium Carbonate), CaMg(CO3)2 (Calcium Magnesium Carbonate) | Solid Solid | Sedimentary Rock, Shells, Sedimentary Rock |
Human Actions Impact the Carbon Cycle
Humans have changed the natural balance of the carbon cycle because we use coal, oil, and natural gas to supply our energy demands. Fossil fuels are a sink for CO2 when they form, but they are a source for CO2 when they are burned.
The equation for combustion of propane, which is a simple hydrocarbon looks like this:
The equation shows that when propane burns, it uses oxygen and produces carbon dioxide and water. So when a car burns a tank of gas, the amount of CO2 in the atmosphere increases just a little. Added over millions of tanks of gas and coal burned for electricity in power plants and all of the other sources of CO2, the result is the increase in atmospheric CO2 seen in the figure above.
The second largest source of atmospheric CO2 is deforestation. Trees naturally absorb CO2 while they are alive. Trees that are cut down lose their ability to absorb CO2. If the tree is burned or decomposes, it becomes a source of CO2. A forest can go from being a carbon sink to being a carbon source.
This forest in Mexico has been cut down and burned to clear forested land for agriculture.
Why the Carbon Cycle is Important
Why is such a small amount of carbon dioxide in the atmosphere even important? Carbon dioxide is a greenhouse gas. Greenhouse gases trap heat energy that would otherwise radiate out into space, which warms Earth.
Summary
- Carbon must be recycled through living organisms or it stays in the atmosphere.
- Carbon cycles quickly between organisms and the atmosphere.
- Due to human activities, there is more carbon dioxide in the atmosphere today than in the past hundreds of thousands of years.
- Carbon is essential for life as part of proteins, carbohydrates, and fats.
- The amount of carbon dioxide in the atmosphere is extremely low, but it is extremely important since carbon dioxide is a greenhouse gas, which helps to keep Earth's climate moderate.
- The amount of carbon dioxide in the atmosphere is rising, a fact that has been documented on Mauna Loa volcano since 1958.