READ: Conduction and Convection
READ: Conduction and Convection
What Is Conduction?
Yummy! These cookies look delicious. But watch out! They just finished baking in a hot oven, so the cookie sheet is too hot to handle without an oven mitt. Touching the cookie sheet with bare hands could cause a painful burn. Do you know why? The answer is conduction.
Conduction is the transfer of thermal energy between particles of matter that are touching. Thermal energy is the total kinetic energy of moving particles of matter, and the transfer of thermal energy is called heat. Conduction is one of three ways that thermal energy can be transferred (the other ways are convection and thermal radiation). Thermal energy is always transferred from matter with a higher temperature to matter with a lower temperature.
Pass It On
To understand how conduction works, you need to think about the tiny particles that make up matter. The particles of all matter are in constant random motion, but the particles of warmer matter have more energy and move more quickly than the particles of cooler matter. When particles of warmer matter collide with particles of cooler matter, they transfer some of their thermal energy to the cooler particles. From particle to particle, like dominoes falling, thermal energy moves through matter.
In the opening photo above, conduction occurs between particles of metal in the cookie sheet and anything cooler that comes into contact with it—hopefully, not someone’s bare hands!
Examples of Conduction
The cookie sheet in the opening image transfers thermal energy to the cookies and helps them bake. There are many other common examples of conduction. The figure below shows a few situations in which thermal energy is transferred in this way.
Hot Iron: A hot iron removes the wrinkles in a shirt. Hot Cocoa: Holding a cup of hot cocoa feels good when you have cold hands. Camp Stove: This camp stove can be used to cook food in a small pot. Snow: Ouch! Can you imagine how cold this snow must feel on bare feet?
Q: How is thermal energy transferred in each of the situations pictured in the figure above?
A: Thermal energy is transferred by conduction from the hot iron to the shirt, from the hot cup to the hand holding it, from the flame of the camp stove to the bottom of the pot as well as from the bottom of the pot to the food inside, and from the feet to the snow. The shirt, hand, pot, food, and snow become warmer because of the transferred energy. Because the feet lose thermal energy, they feel colder.
What is Convection?
Do you see the water bubbling in this pot? The water is boiling hot. How does all of the water in the pot get hot when it is heated only from the bottom by the gas flame? The answer is convection.
Convection is the transfer of thermal energy by particles moving through a fluid (either a gas or a liquid). Thermal energy is the total kinetic energy of moving particles of matter, and the transfer of thermal energy is called heat. Thermal energy is always transferred from matter with a higher temperature to matter with a lower temperature.
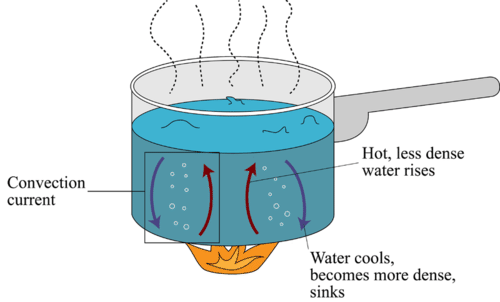
How Does Convection Occur?
The figure below shows how convection occurs, using hot water in a pot as an example. When particles in one area of a fluid (in this case, the water at the bottom of the pot) gain thermal energy, they move more quickly, have more collisions, and spread farther apart. This decreases the density of the particles, so they rise up through the fluid. As they rise, they transfer their thermal energy to other particles of the fluid and cool off in the process. With less energy, the particles move more slowly, have fewer collisions, and move closer together. This increases their density, so they sink back down through the fluid. When they reach the bottom of the fluid, the cycle repeats. The result is a loop of moving particles called a convection current.
Examples of Convection
Convection currents transfer thermal energy through many fluids, not just hot water in a pot. For example, convection currents transfer thermal energy through molten rock below Earth’s surface, through water in the oceans, and through air in the atmosphere. Convection currents in the atmosphere create winds.
Summary
- Conduction is the transfer of thermal energy between particles of matter that are touching. Thermal energy is always transferred from particles of warmer matter to particles of cooler matter.
- When particles of warmer matter collide with particles of cooler matter, they transfer some of their thermal energy to the cooler particles.
- Convection is the transfer of thermal energy by particles moving through a fluid. Thermal energy is always transferred from an area with a higher temperature to an area with a lower temperature.
- Moving particles transfer thermal energy through a fluid by forming convection currents.
- Convection currents move thermal energy through many fluids, including molten rock inside Earth, water in the oceans, and air in the atmosphere.